Chemistry 100 Review of Charles and Boyles Laws
five.3: The Simple Gas Laws- Boyle's Law, Charles's Police force and Avogadro'due south Law
- Page ID
- 37996
Learning Objectives
- To understand the relationships among pressure, temperature, volume, and the amount of a gas.
Early on scientists explored the relationships among the force per unit area of a gas (P) and its temperature (T), volume (V), and amount (n) by holding two of the four variables abiding (amount and temperature, for case), varying a third (such as pressure), and measuring the result of the change on the fourth (in this case, book). The history of their discoveries provides several excellent examples of the scientific method.
The Relationship between Pressure and Volume: Boyle's Police force
Equally the pressure level on a gas increases, the book of the gas decreases because the gas particles are forced closer together. Conversely, every bit the pressure on a gas decreases, the gas volume increases considering the gas particles can now move farther apart. Weather balloons go larger as they rise through the temper to regions of lower pressure considering the volume of the gas has increased; that is, the atmospheric gas exerts less pressure on the surface of the balloon, so the interior gas expands until the internal and external pressures are equal.
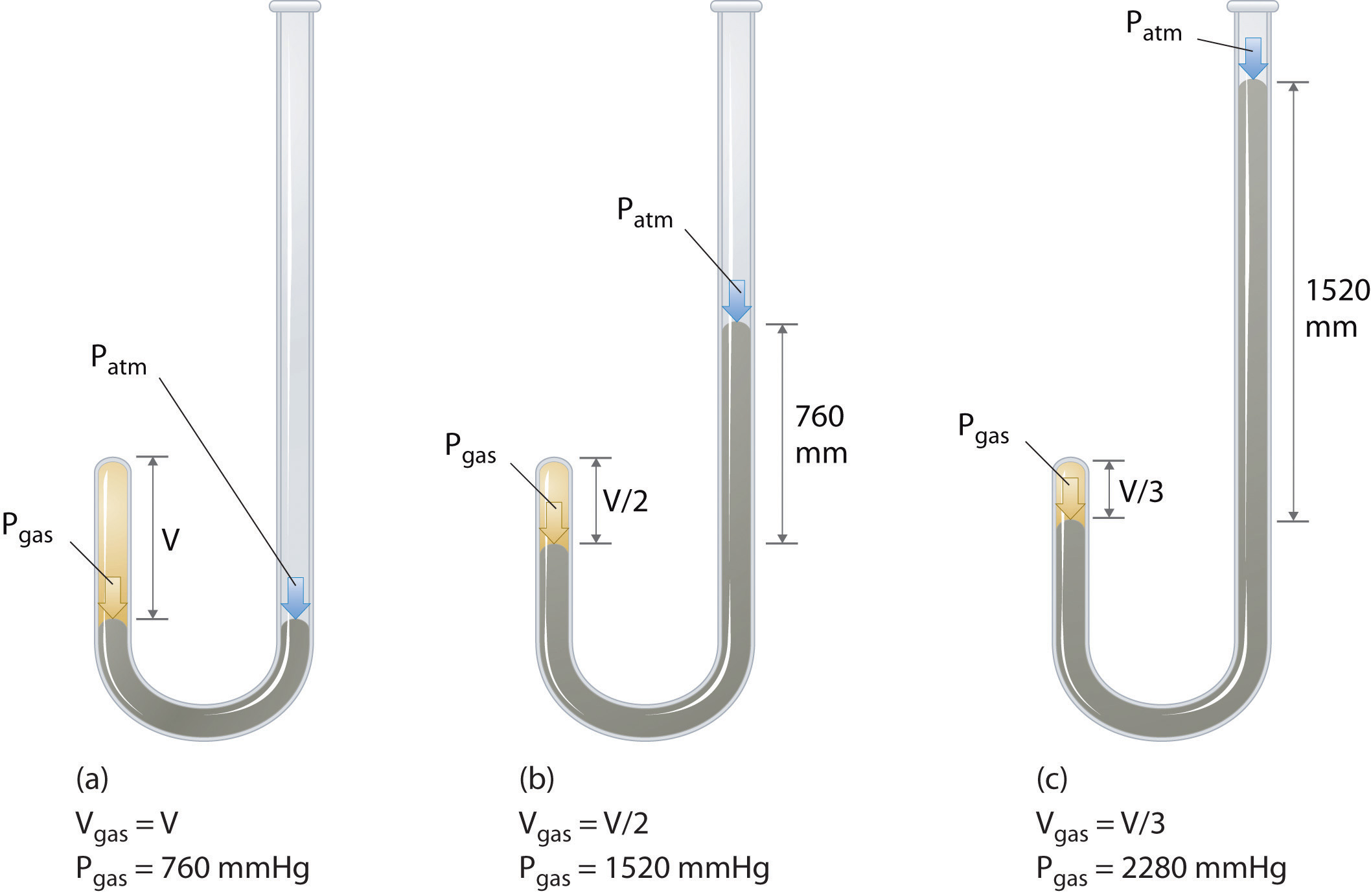
The Irish chemist Robert Boyle (1627–1691) carried out some of the earliest experiments that determined the quantitative human relationship betwixt the pressure and the volume of a gas. Boyle used a J-shaped tube partially filled with mercury, every bit shown in Figure \(\PageIndex{1}\). In these experiments, a small amount of a gas or air is trapped above the mercury column, and its volume is measured at atmospheric pressure and abiding temperature. More than mercury is and so poured into the open arm to increase the pressure on the gas sample. The pressure on the gas is atmospheric pressure plus the difference in the heights of the mercury columns, and the resulting volume is measured. This process is repeated until either at that place is no more room in the open arm or the volume of the gas is too small to exist measured accurately. Data such as those from one of Boyle'southward ain experiments may be plotted in several ways (Figure \(\PageIndex{two}\)). A uncomplicated plot of \(V\) versus \(P\) gives a curve called a hyperbola and reveals an changed relationship between pressure level and volume: as the pressure is doubled, the book decreases by a factor of two. This relationship between the two quantities is described equally follows:
\[PV = \rm abiding \label{10.3.1}\]
Dividing both sides past \(P\) gives an equation illustrating the changed relationship between \(P\) and \(V\):
\[V=\dfrac{\rm const.}{P} = {\rm const.}\left(\dfrac{i}{P}\correct) \characterization{10.3.2}\]
or
\[V \propto \dfrac{1}{P} \label{10.3.3}\]
where the ∝ symbol is read "is proportional to." A plot of 5 versus ane/P is thus a direct line whose gradient is equal to the constant in Equations \(\ref{10.3.one}\) and \(\ref{10.3.3}\). Dividing both sides of Equation \(\ref{10.iii.1}\) by V instead of P gives a similar relationship between P and i/Five. The numerical value of the abiding depends on the amount of gas used in the experiment and on the temperature at which the experiments are carried out. This human relationship between pressure level and volume is known equally Boyle'south police, afterward its discoverer, and can exist stated as follows: At constant temperature, the volume of a stock-still amount of a gas is inversely proportional to its pressure. This law in practice is shown in Figure \(\PageIndex{2}\).
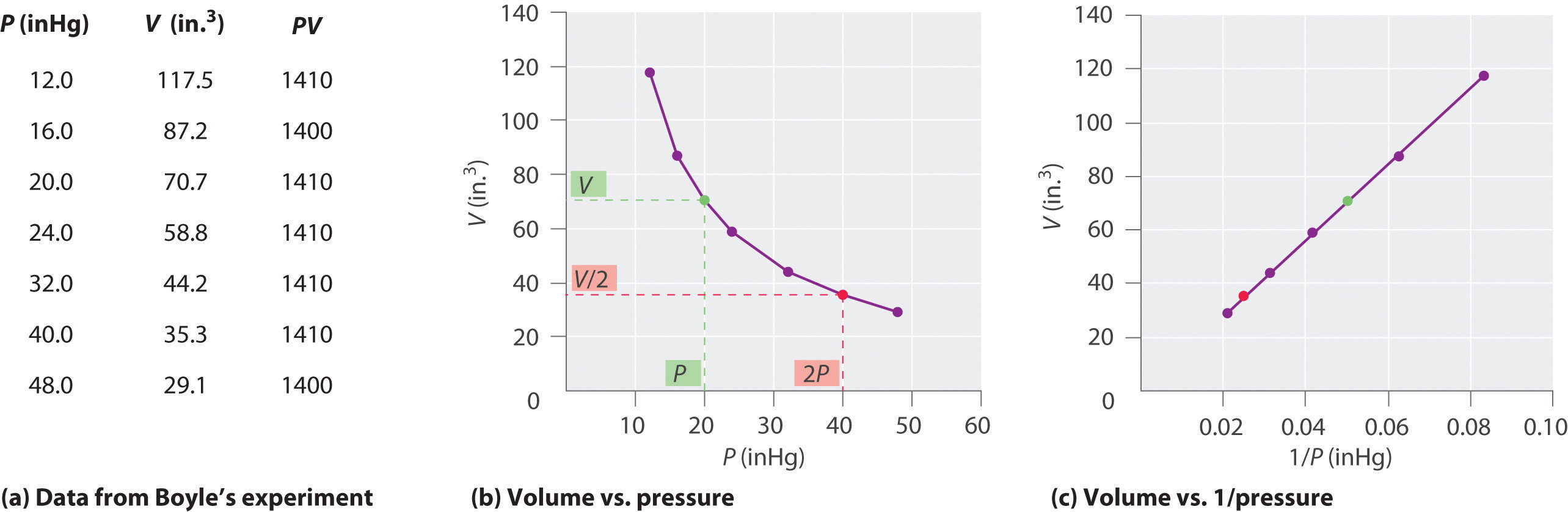
At abiding temperature, the volume of a stock-still corporeality of a gas is inversely proportional to its pressure level
The Human relationship between Temperature and Volume: Charles's Police
Hot air rises, which is why hot-air balloons ascend through the temper and why warm air collects near the ceiling and cooler air collects at ground level. Because of this behavior, heating registers are placed on or near the floor, and vents for air-conditioning are placed on or almost the ceiling. The key reason for this behavior is that gases expand when they are heated. Considering the same amount of substance now occupies a greater volume, hot air is less dense than cold air. The substance with the lower density—in this case hot air—rises through the substance with the college density, the cooler air.
The first experiments to quantify the relationship betwixt the temperature and the volume of a gas were carried out in 1783 past an gorging balloonist, the French chemist Jacques Alexandre César Charles (1746–1823). Charles's initial experiments showed that a plot of the volume of a given sample of gas versus temperature (in degrees Celsius) at constant pressure is a straight line. Similar but more than precise studies were carried out past another balloon enthusiast, the Frenchman Joseph-Louis Gay-Lussac (1778–1850), who showed that a plot of V versus T was a straight line that could exist extrapolated to a indicate at zero volume, a theoretical condition now known to correspond to −273.15°C (Figure \(\PageIndex{iii}\)).A sample of gas cannot really have a volume of nada considering whatsoever sample of matter must accept some volume. Furthermore, at ane atm force per unit area all gases liquefy at temperatures well to a higher place −273.fifteen°C. Annotation from part (a) in Effigy \(\PageIndex{iii}\) that the slope of the plot of V versus T varies for the aforementioned gas at dissimilar pressures but that the intercept remains constant at −273.xv°C. Similarly, as shown in office (b) in Figure \(\PageIndex{three}\), plots of V versus T for dissimilar amounts of varied gases are straight lines with unlike slopes but the same intercept on the T axis.
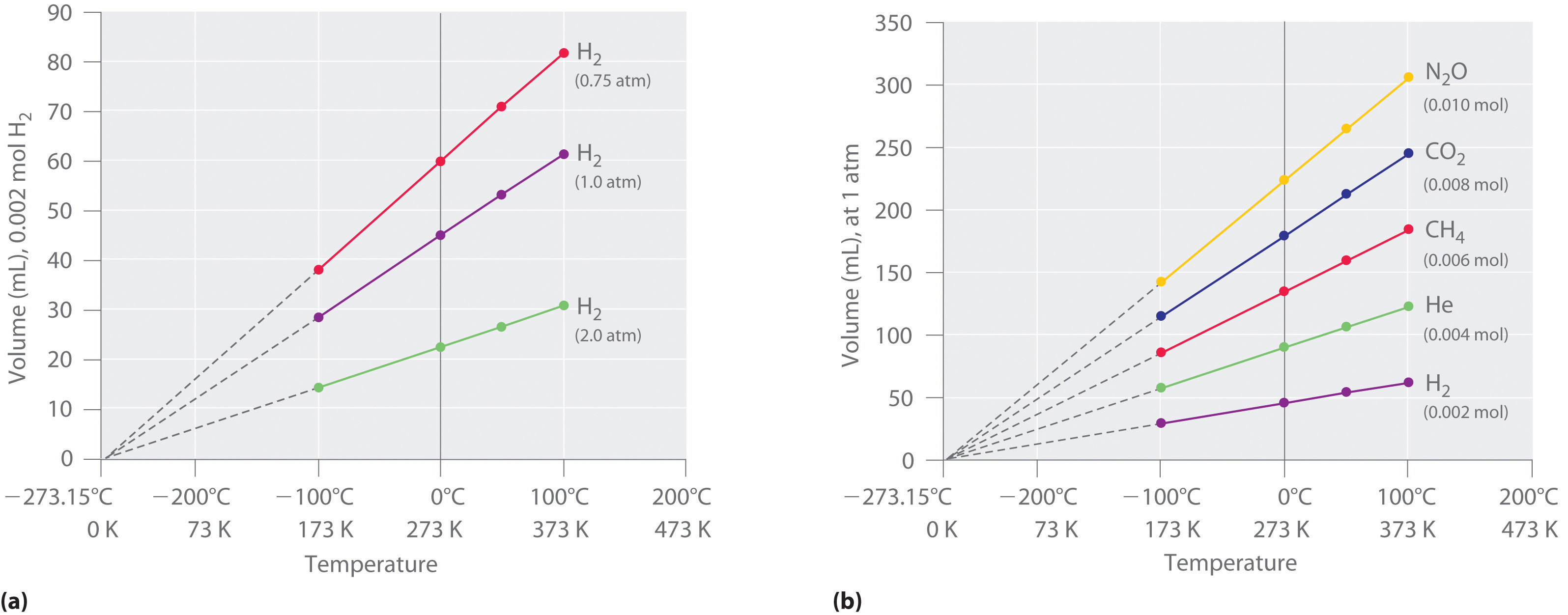
The significance of the invariant T intercept in plots of 5 versus T was recognized in 1848 by the British physicist William Thomson (1824–1907), later named Lord Kelvin. He postulated that −273.15°C was the lowest possible temperature that could theoretically exist accomplished, for which he coined the term absolute zero (0 K).
We can country Charles'south and Gay-Lussac's findings in simple terms: At abiding pressure level, the volume of a stock-still corporeality of gas is directly proportional to its absolute temperature (in kelvins). This relationship, illustrated in part (b) in Figure \(\PageIndex{3}\) is often referred to as Charles's law and is stated mathematically as
\[5 ={\rm const.}\; T \label{10.3.iv}\]
or
\[V \propto T \label{10.3.5}\]
with temperature expressed in kelvins, not in degrees Celsius. Charles's law is valid for most all gases at temperatures well higher up their humid points.
The Human relationship between Corporeality and Volume: Avogadro'due south Law
We can demonstrate the relationship between the volume and the corporeality of a gas past filling a balloon; as we add more than gas, the balloon gets larger. The specific quantitative human relationship was discovered past the Italian chemist Amedeo Avogadro, who recognized the importance of Gay-Lussac'due south work on combining volumes of gases. In 1811, Avogadro postulated that, at the same temperature and pressure, equal volumes of gases contain the same number of gaseous particles (Figure \(\PageIndex{iv}\)). This is the historic "Avogadro's hypothesis."
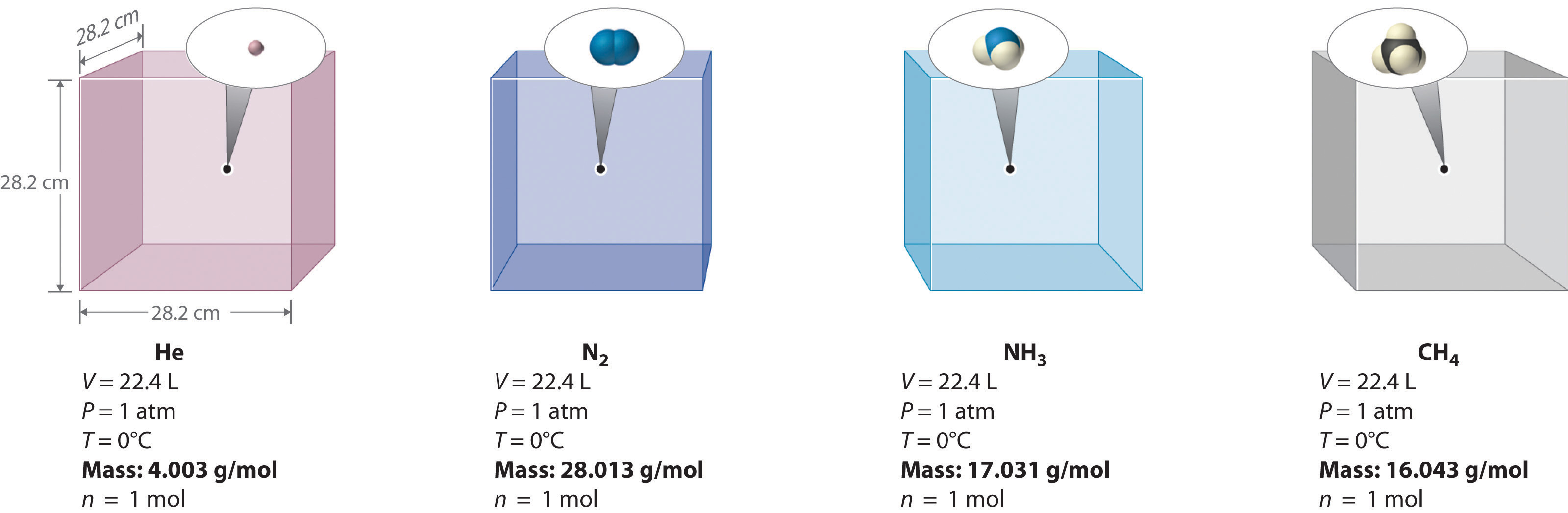
A logical corollary to Avogadro'southward hypothesis (sometimes called Avogadro'south law) describes the human relationship between the volume and the amount of a gas: At abiding temperature and pressure, the volume of a sample of gas is directly proportional to the number of moles of gas in the sample. Stated mathematically,
\[V ={\rm const.} \; (north) \label{10.3.6}\]
or
\[V \propto.n \text{@ constant T and P} \label{10.3.7}\]
This relationship is valid for most gases at relatively low pressures, but deviations from strict linearity are observed at elevated pressures.
For a sample of gas,
- V increases as P decreases (and vice versa)
- V increases as T increases (and vice versa)
- V increases as n increases (and vice versa)
The relationships among the book of a gas and its pressure, temperature, and corporeality are summarized in Effigy \(\PageIndex{v}\). Volume increases with increasing temperature or amount, but decreases with increasing force per unit area.
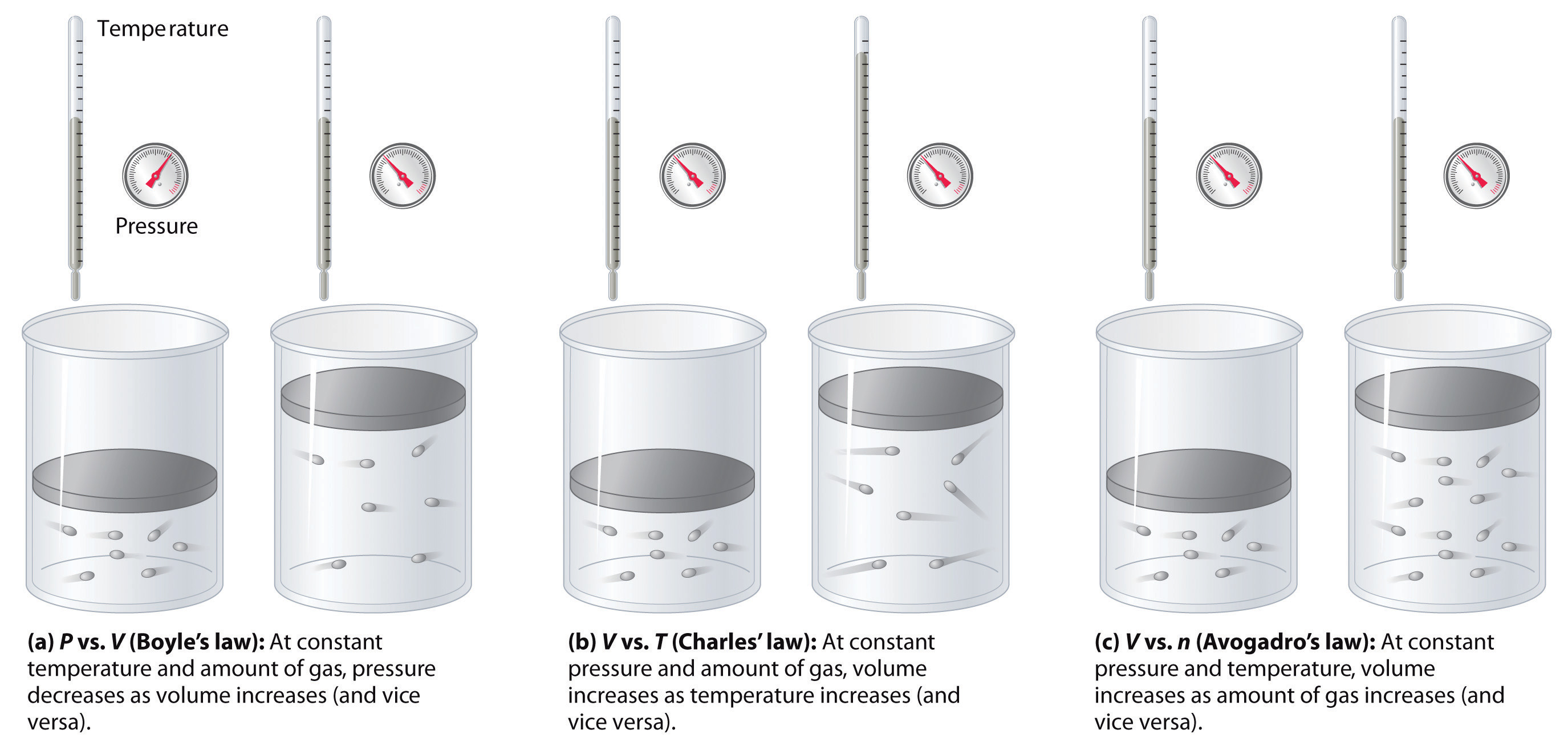
Summary
The volume of a gas is inversely proportional to its pressure and direct proportional to its temperature and the amount of gas. Boyle showed that the volume of a sample of a gas is inversely proportional to its force per unit area (Boyle's law), Charles and Gay-Lussac demonstrated that the volume of a gas is directly proportional to its temperature (in kelvins) at constant pressure (Charles'due south police), and Avogadro postulated that the volume of a gas is directly proportional to the number of moles of gas nowadays (Avogadro's constabulary). Plots of the volume of gases versus temperature extrapolate to zilch volume at −273.xv°C, which is absolute cypher (0 K), the everyman temperature possible. Charles's police implies that the volume of a gas is directly proportional to its accented temperature.
Source: https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_A_Molecular_Approach_(Tro)/05%3A_Gases/5.03%3A_The_Simple_Gas_Laws-_Boyles_Law_Charless_Law_and_Avogadros_Law
إرسال تعليق for "Chemistry 100 Review of Charles and Boyles Laws"